Attributes related to lyophilization of glass containers
As an important member of pharmaceutical packaging
materials, glass packaging has withstood the test of time, meeting the vast
majority of requirements from the early stages of drug development (such as
physical and chemical properties, pharmaceutical compatibility, etc.), through
production scale-up (process adaptability, etc.), to clinical trials and market
launch (safety, etc.). Despite its long history, however, there is still a lack
of understanding about glass packaging materials among pharmaceutical
professionals or packaging professionals to some extent. Therefore, we will
share knowledge with you based on industry literature and reviews, hoping it
will be helpful to everyone.
I. The lyophilization process of pharmaceuticals
In the production process of pharmaceuticals, if it involves
lyophilized powder injections, the lyophilization process will be involved.
Before discussing the correlation between the attributes of glass packaging and
the lyophilization process, we first need to understand what lyophilization
process and products are, as shown in Figure 1. As you can see, the process can
be simplified into three steps: after the liquid medicine is lyophilized, the
rubber stopper is in a semi-inserted state (to provide a channel for moisture
to evaporate outward). In Stage I, unbound free water molecules are removed,
and then the temperature is further increased to remove bound water molecules
(bound through hydrogen bonds, etc.) in Stage II. After that, inert gas is
reintroduced to complete the stoppering process.

Figure 1: Lyophilized Products and Related Process
II. Glass attributes related to lyophilization
After understanding the lyophilization
process mentioned earlier, let's delve into the glass attributes related to
lyophilization. There are five aspects as follows:
(1) Coefficient of Thermal Expansion (CTE).
From a materials science perspective, it
characterizes the volume change of a material when heated or cooled, and its
value is related to the glass composition and the measurement range. The
smaller this value, the more stable the material is during temperature changes.
Strictly speaking, CTE can be further divided into linear expansion, areal
expansion, and volumetric expansion, and the essence of these is the increase
in the gaps between the atoms material. Pharmaceutical glass packaging is
mostly made of borosilicate glass, with a CTE of approximately 3-10**10-6K-1.
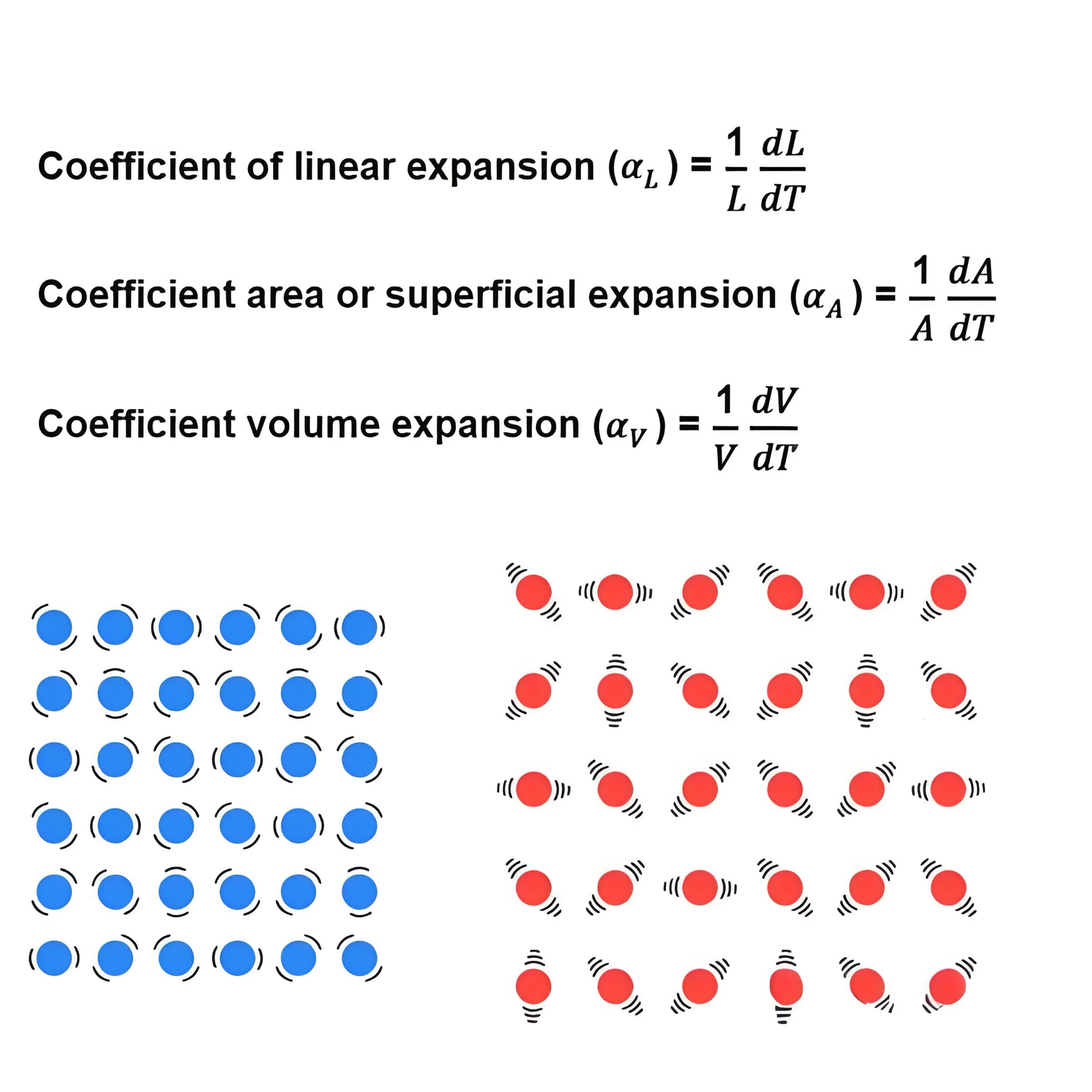
Figure 2: Coefficient of Expansion and Its
Principle
(2) Thermal conductivity
This is an indicator of a material's thermal
conduction performance. Different media have different mechanisms for heat
conduction. For example, metals rely on electrons, nonmetals rely on lattice
structure vibrations, and gases rely on molecular thermal motion. For glass
materials, their thermal conductivity is low. If glass packaging containers are
involved in temperature changes in pharmaceutical manufacturing, such as
cleaning empty bottles to remove pyrogens or product lyophilization, this
property becomes important. Generally, glass materials are similar, so the only
significant difference lies in the impact of the product's geometric design on
heat conduction.
(3) Residual Stress
Residual stress refers to the self-balancing internal stress that
remains in an object after the elimination of external forces or uneven
temperature fields. For example, if a spring is subjected to a compressive or
tensile force, ideally, after the force is removed, it has a driving force to
return to its original state. However, if the recovery process is particularly
slow after the external force is removed, this force will persist for some
time, known as residual stress. For glass, during the processing and refining
stages, such as melting and shaping, when the heat source is removed, the
movement of atoms is not as smooth as it is at high temperatures. This force
trying to return to the original state is residual stress. The residual stress
in glass can be revealed using polarized light. Glass is an isotropic material
with the same refractive index in all directions. If there is stress in the
glass, the isotropic nature is disrupted, causing changes in the refractive
index. The refractive indices in the two principal stress directions are no
longer the same, leading to birefringence.
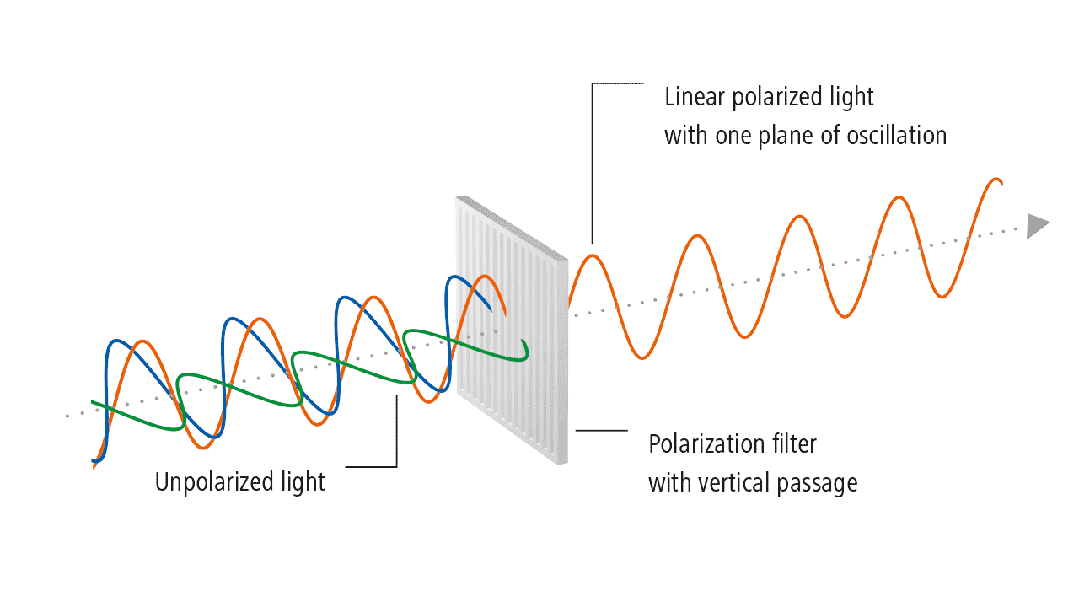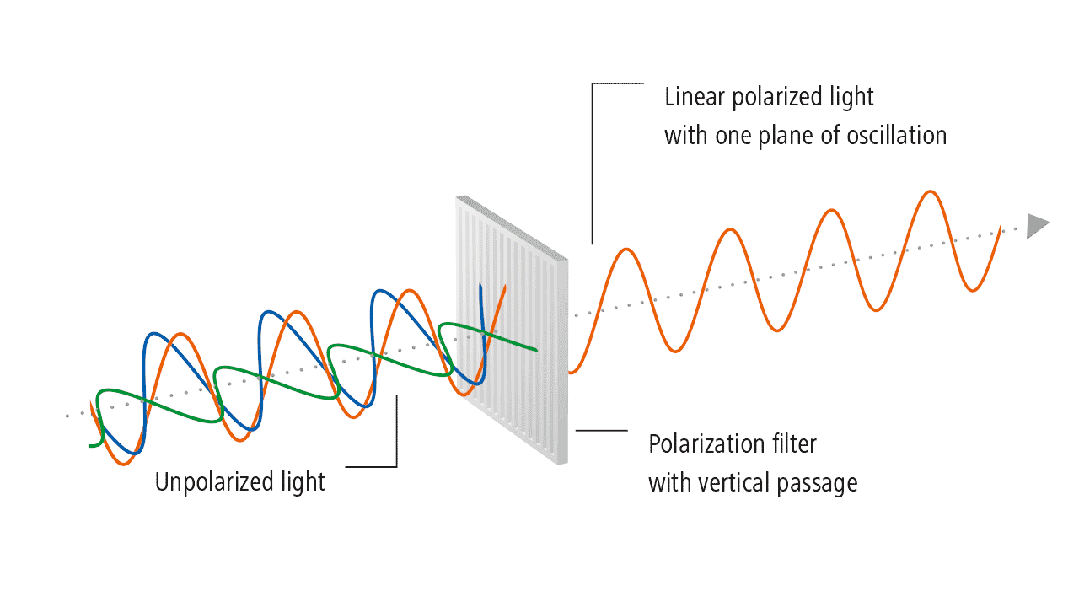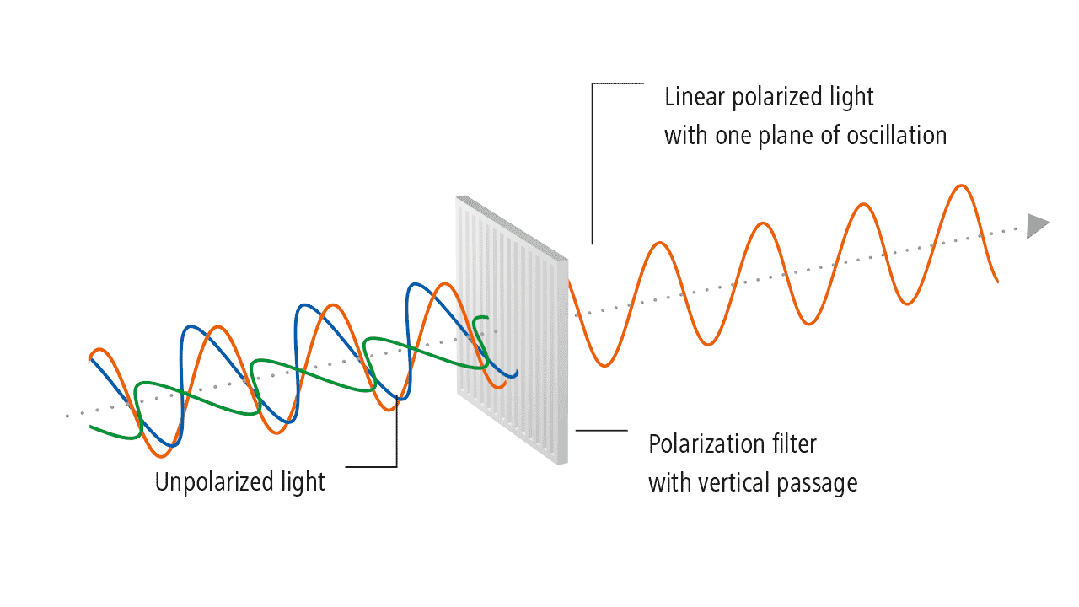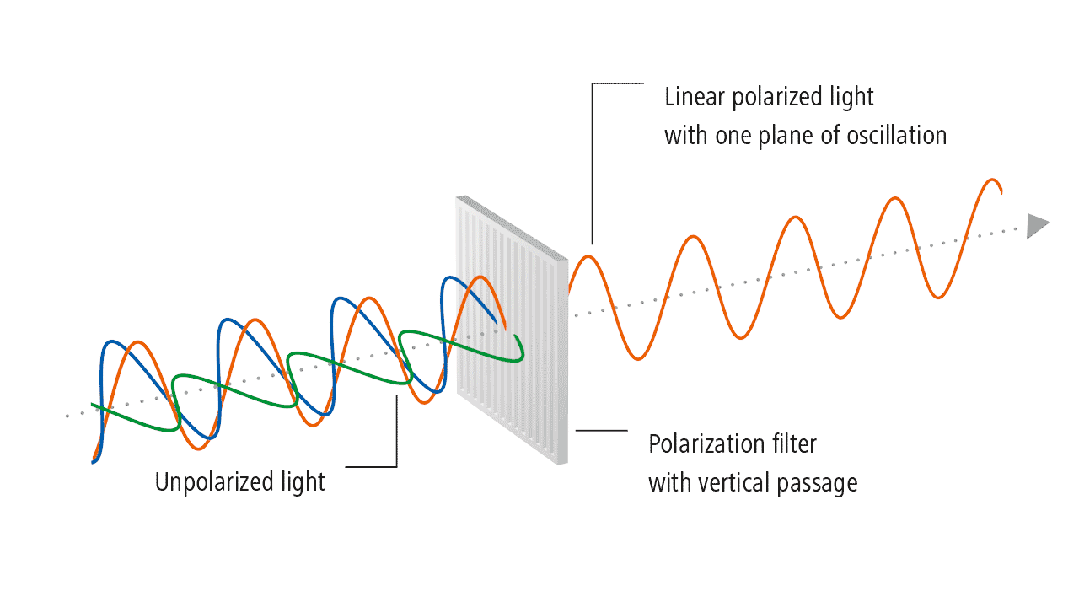

Figure 3: Stress Detection Process
(4) Thermal shock resistance
This indicator is actually a combined result
of the expansion coefficient, packaging container design, and stress. It
measures the ability of the packaging container to withstand temperature
changes, such as transitioning from high temperature to room temperature or
from room temperature to high temperature. Due to different glass materials
having varying expansion coefficients, and glass being a poor conductor of
heat, theoretically, the smaller the expansion coefficient, the stronger its
ability to withstand thermal shock under the same thickness conditions. This is
why borosilicate glass has a stronger resistance to thermal shock than
soda-lime glass.

Figure 4: Thermal expansion caused by temperature gradient
(5) Crack resistance
Crack resistance refers more to the ability to withstand cracking due to mechanical forces. Its strength is related to the design of the container, the type of material, and whether there are defects on the surface. Sometimes, the cracking of glass bottles is caused by mechanical forces, such as expansion during freeze-drying.

Figure 5: Cracking caused by expansion during the freeze-drying process
III. Parameters related to cracking during freeze-drying
The characteristics of lyophilized
preparations are that the glass packaging material undergoes thermal shock,
while the internal formulation undergoes a process of cooling and expansion.
Therefore, the cracking during the lyophilization process may primarily be due
to the following reasons:
◇ The process and design of glass packaging
materials: Sometimes, the entire bottom of the bottle may fall off, which may
be related to insufficient annealing of the bottle and the fragile edge of the
bottom molding.
◇ Formulation-related: Sometimes certain
special ingredients in the formulation may tend to cause excessive expansion.
◇ Filling volume: Sometimes the container is
overfilled, leaving insufficient space for freezing expansion during the
lyophilization process.
◇ Local defects in glass packaging during the
manufacturing process: Defects become the starting point for cracking.小结
Ⅳ. Conclusion
◇ The lyophilization performance of glass
packaging is related to many attributes.
◇ Cracking during the lyophilization process is related to the material,
design of the glass packaging, and the formulation of the product.
Post time: 2025-03-12

